Sp-one/p 316271-One punch man sp
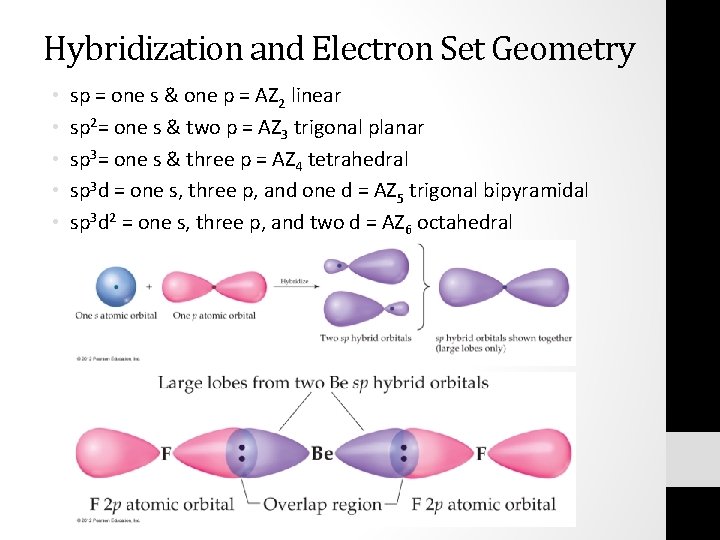
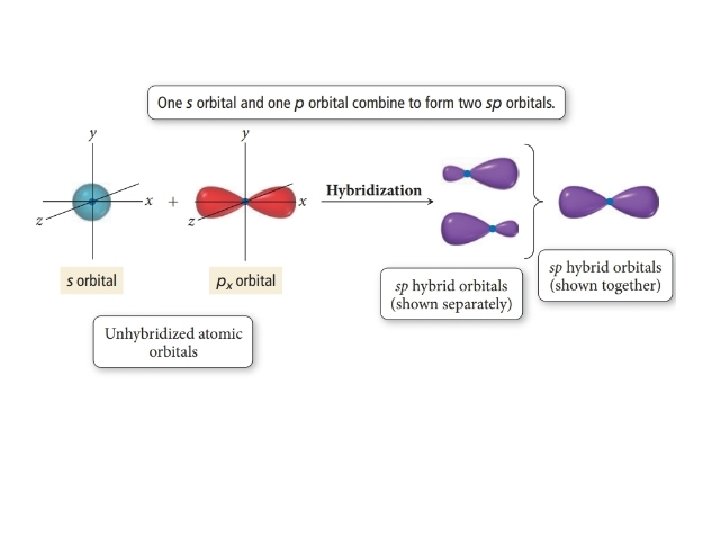
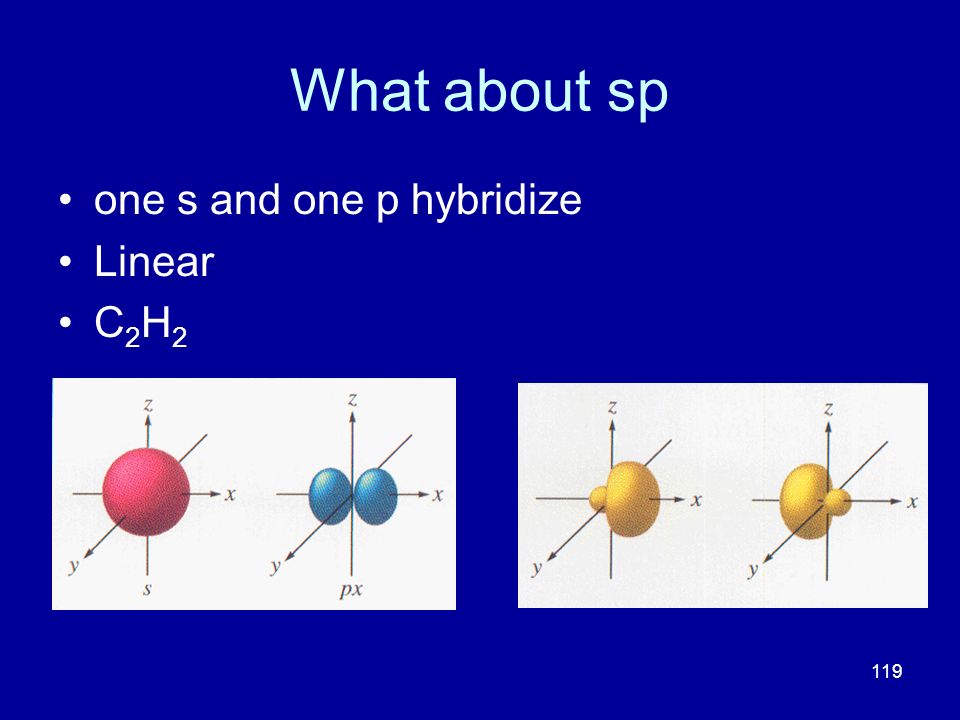
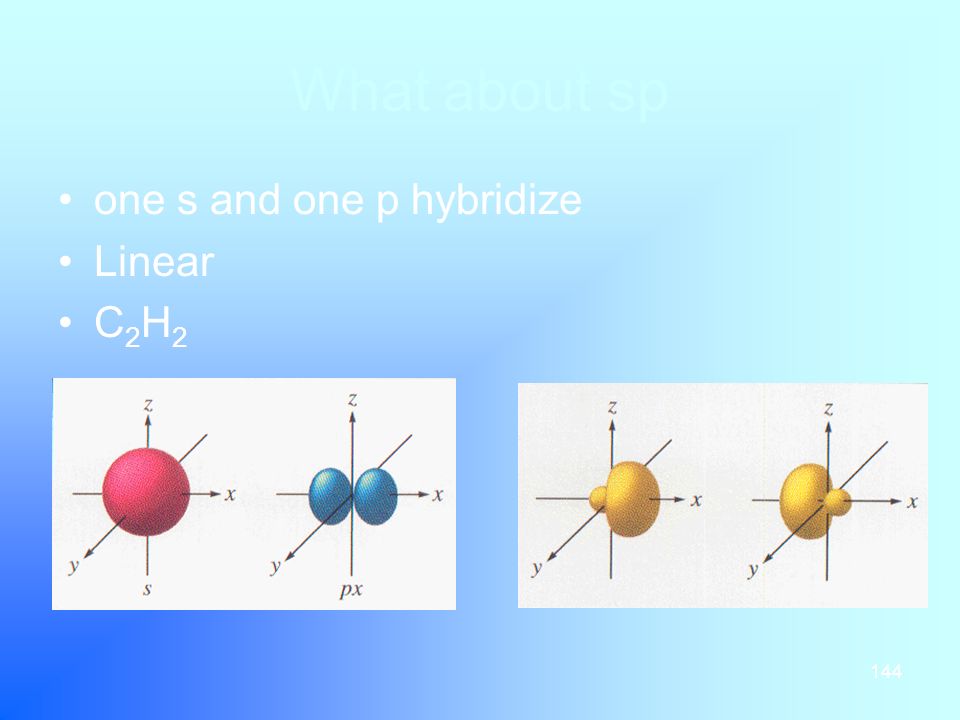
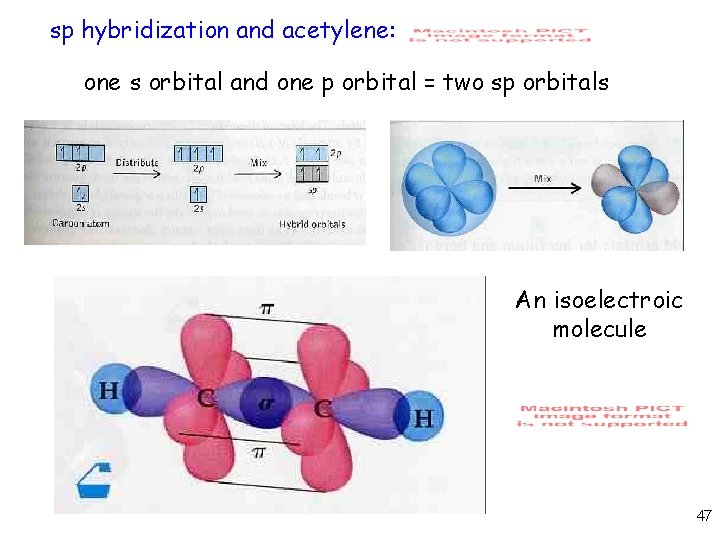
Sp hybridization (mixing of one s orbital and one p orbital) Sp Orbitals Mixing the s and p orbitals yields two degenerate orbitals that are hybrids of the two orbitals The sp hybrid orbitals each have two lobes like a p orbital One of the lobes is larger and more rounded, as is the s orbital Position of sp Orbitals These two degenerate orbitals would align themselves 180° from eachThe Game Boy Advance SP (GBA SP), released in Japan on , is a sixthgeneration handheld game console developed, released, and marketed by Nintendo that served as an upgraded version of the original Game Boy AdvanceThe "SP" in the name stands for "Special" It is the penultimate console in the Game Boy Advance family before the Game Boy Micro, released inAn sp hybridized atom uses one s and one p orbital to make two sp hybrid orbitals;
Plos One Seed Biopriming With P And K Solubilizing Enterobacter Hormaechei Sp Improves The Early Vegetative Growth And The P And K Uptake Of Okra Abelmoschus Esculentus Seedling
One punch man sp
One punch man sp-Sign In Sign in with your organizational account Look up your email address / student ID Reset Password Create PasswordFriday marked day one of a market rally attempt, but it's still a correction Tesla deliveries jumped Tesla delivered more than 240,000 cars in the third quarter of 21 Wall Street was looking




Spone
S&P Capital IQ Pro New User Sign Up Ratings360 ® New User Sign Up Platts New User Sign Up MarketPlace New User Sign UpGo to the Windows 7 Service Pack 1 download page on the Microsoft website Select Install Instructions to see which packages are available for download, and make note of the one that you need Select the appropriate language from the dropdown list, and then select Download Select the packages you need to install, select Next, and then followSp 2 Hybridization The valence orbitals of a central atom surrounded by three regions of electron density consist of a set of three sp 2 hybrid orbitals and one unhybridized p orbital This arrangement results from sp 2 hybridization, the mixing of one s orbital and two p orbitals to produce three identical hybrid orbitals oriented in a trigonal planar geometry (Figure
In the case you describe, mixing one s atomic orbital with two p atomic orbitals means that three hybrid atomic orbitals must result These three hybrid orbitals will each contain % s character and % p character because that is the ratio of the one s orbital to two p orbitals that we are mixing togetherControversial Opinion This Time is one of SP's top 10 songs Discussion Close Vote Posted by Mellon Collie and the Infinite Sadness 5 minutes ago but I was lucky enough to snag one of the two JMP1 preamps from the Reverb sale It cost a little over 5k and I'm not rich or even upper middle class by any stretch but I've been one ofS&P 500 PE Ratio chart, historic, and current data Current S&P 500 PE Ratio is 3360, a change of 040 from previous market close
P 22 12 L E M o U o L M LIN co s L M 14 —i E c žs A GIO M o sp 961 PLATE 3 OF 14 Oil and gas production from the Grove Sandstone Charles J Mankin, Director 24 EXPLANATION Oil well o Gas well Based on NRIS wellfile data, compiled from operatorreported well completions (Oklahoma Corporation Commission forms 1002A) 25 25 27 A w A 27 25Question The blending of one s atomic orbital and one pSP 951 P 6TIF Author murr5940 Created Date PM




One Lockheed P2v 7b P 2h Sp 2h Neptune Maritime Patrouille Plane 1961 19 On Flykamp Valkenburg Stock Photo Alamy




A Sp Hybridisation One S Orbital Mixes With One P Orbital Forming Two Equivalent Sp Hybrid Orbitals Types Of Hybridisation Linear 180 Two Sp Hybrid
Bringing an app store experience to IU servicesTwo sp orbitals will be at 180 degrees to each other Any central atom surrounded by just two regions of valence electron density in a molecule will exhibit sp hybridization Some examples include the mercury atom in the linear HgCl 2 molecule (The notation sp^2 comes from mixing one s and two p orbitals together to get 33% s character and 66% p character) Hence, the \mathbf(sigma) bonds of ethene (shown in yellow) lie on a single plane In addition, we have the \mathbf(2p_z) atomic orbital (perpendicular to the plane of the molecule, in purple) from each carbon that is used to




Winchester 30 06 180 Gr Power Point Sp One Full One Partial 35 Rounds




Cven 5424 Environmental Organic Chemistry Lecture 2 Organic Chemistry And Thermodynamics Ppt Download
Current and Historical Performance Performance for SPDR S&P 500 on Yahoo FinanceSp 2 Hybridization The valence orbitals of a central atom surrounded by three regions of electron density consist of a set of three sp 2 hybrid orbitals and one unhybridized p orbital This arrangement results from sp 2 hybridization, the mixing of one s orbital and two p orbitals to produce three identical hybrid orbitals oriented in a trigonal planar geometry ()Index performance for S&P 500 INDEX (SPX) including value, chart, profile & other market data




One Lockheed P2v 7b P 2h Sp 2h Neptune Maritime Patrouille Plane 1961 19 On Flykamp Valkenburg Stock Photo Alamy



Page 3 6 April Calendar High Resolution Stock Photography And Images Alamy
Open SAP ONE Support Launchpad;The unhybridized atomic p orbital lies at a 90° angle to the plane This configuration allows for the maximum separation of all orbitals Last, the atomic orbitals of carbon can hybridize by the linear combination of one s and one p orbital This process forms two equivalent sp hybrid orbitals The remaining two atomic p orbitals remainFor the hydrogen fluoride molecule, for example, two F lone pairs are essentially unhybridized p orbitals, while the other is an sp x hybrid orbital An analogous consideration applies to water (one O lone pair is in a pure p orbital, another is in an sp x hybrid orbital) See also Crystal field theory;



Chapter 9 Notes Ap Chemistry Galster Molecular Geometry



Which Of The Following Hybridization Schemes Allows Chegg Com
In sp hybridization, the s orbital overlaps with only one p orbital Atoms that exhibit sp hybridization have sp orbitals that are linearly oriented;Celsus Sound SPONE/P 小型ブックシェルフ型スピーカー NEW 定価60,000円(ペア/税別) 形式 : 2ウェイ・バスレフ型 搭載ユニット ・高域 : ソフトドーム型 ・低域 : 35インチ・カーボンファイバー・コーン 周波数特性 : 50Hz~kHzYou are now logged out of the SAP ONE Support Launchpad What would you like to do next?




Secrets Are Deadly Kindle Edition By Mowre S P Mystery Thriller Suspense Kindle Ebooks Amazon Com




Arena Sp Der One P Ece Girls Swimwear Superdokan
『spone/p』の価格を超えた仕上げにマッチしています。 縦置きにも対応しており、省スペースでのシステム構築にも有利です。 ※YAMAHA WXA50 縦置き時Best LowLatency Data Feed Provider;Sp 3 Hybridization When one 's' orbital and 3 'p' orbitals belonging to the same shell of an atom mix together to form four new equivalent orbital, the type of hybridization is called a tetrahedral hybridization or sp 3 The new orbitals formed are called sp 3 hybrid orbitals



Plos One Seed Biopriming With P And K Solubilizing Enterobacter Hormaechei Sp Improves The Early Vegetative Growth And The P And K Uptake Of Okra Abelmoschus Esculentus Seedling



Chapter 10 Chemical Bonding Molecular Shapes Valence Bond
ESG is one of our biggest growth opportunities, S&P Dow Jones CEO says Environmental, social and governance investing is making a big splash in the world of indexing BlackRock's iShares hasThus, the ethylene molecule is said to have sp2s bonds (4 CH bonds) , one sp 2sp 2 bond (CC bond) and one pp pi bond (CC bond) Thus, the sp 2 hybridization theory explains the double bond, the trigonal planar structure in ethylene molecule Similarly, for a triple bond formation, like for that of acetylene molecule, there is spSp hybridization In sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character This type of hybridization is required whenever an atom is surrounded by two groups of electrons Created by Jay This is the currently selected item




5 The Sp 2 Orbitals Are Formed From One S And Two P Orbitals One P Download Scientific Diagram




Poland S Pzl Gull Wing Fighters Volume One P 1 Through P 8 Warren A Eberspacher Jan P Koniarek Amazon Com Books
My Account, Plan Participation, Investment Funds, Planning and Tools, Life Events andIt is one of the most commonly followed equity indices As of , more than $46 trillion was invested in assets tied to the performance of the index The S&P 500 index is a freeEquivalent sp2 orbitals, leaving one p orbital untouched The process is shown below 2s 2p X 2p y 2p z Potential energy sp2 hybridization sp2 sp2 sp2 p In this top view, the unhybridized p orbital cannot be seen because it also arranges itself to be as far apart from the sp2 orbitals as possible That is to say, it is positioned at right




Lecture 16 C 1403 October 31 05 18




Digi One Sp 1port Device Server Pn 01 For Sale Online Ebay
Sp2 hybridization in ethene In sp^2 hybridization, the 2s orbital mixes with only two of the three available 2p orbitals, forming a total of three sp^2 orbitals with one porbital remaining The two carbon atoms form a sigma bond in the molecule by overlapping two sp 2 orbitals Each carbon atom forms two covalent bonds with hydrogen by s–spEach sp hybrid orbital has one large and one small lobe Hybrid orbitals are designated by using a superscript to indicate the _____ of each type of orbital used to form them For example, the designation sp^2 indicates that _____ s and _____ p orbital(s) were combined to form this orbital typeVisit SAP Support Portal or SAPcom



1 Chemical Bonding 2 Lewis Theory An Overview Valence E Play A Fundamental Role In Chemical Bonding E Transfer Leads To Ionic Bonds Sharing Of Ppt Download




E Mini S Amp P 500 Index Es Futures Technical Analysis 039 One And Done 039 Sell Off Or Actual Change In Trend
On each carbon, one sp hybrid bonds to a hydrogen and the other bonds to the other carbon atom, forming the σ bond skeleton of the molecule In addition to the sp hybrids, each carbon atom has two halfoccupied p orbitals oriented at right angles to each other and to the interatomic axis These two sets of parallel and adjacent p orbitals canThe blending of one s atomic orbital and one p atomic orbital produces _____ a two sp hybrid orbitals b two sp2 hybrid orbitals c three sp2 hybrid orbitals d two sp3 hybrid orbitals e three sp hybrid orbitals;Sp Hybridization In the case of the sp hybridization, only one s and one porbital are mixed together to make hybrids This leaves two unused porbitals The unused sp orbitals force the structure to have a linear 3D geometry The unused porbitals can make two double bonds, a triple bond, or potentially participate in resonance with other




Bookshelf Speakers Review Shootout Celsus Sound Sp One Vs Centrance Masterclass 2504 Vs John Blue Jb3 Headphone Reviews And Discussion Head Fi Org



Plos One Expression Of A Chimeric Antigen Receptor In Multiple Leukocyte Lineages In Transgenic Mice
If your company has a current subscription with S&P Global Market Intelligence, you can register as a new user for access to the platform(s) covered by your license at S&P Capital IQ Pro or S&P Capital IQThere are two remaining p orbitals Next we show the phase pictures of combining the sp hybrid orbitals with fi rst one and then both of the remaining p orbitals Phase pictures loop Diagram Axes for you to draw the loop diagram You will also see these orbitals Home to the S&P 500® and Dow Jones Industrial Average®, S&P Dow Jones Indices is the world's leading resource for benchmarks and investable indices




A Catalogue Of The Collection Of Cambrian And Silurian Fossils Contained In The Geological Museum Of The University Of Cambridge Paleontology Paleontology 42 Upper Cambrian Case And Column Of I Rawers Gk



Plos One Seed Biopriming With P And K Solubilizing Enterobacter Hormaechei Sp Improves The Early Vegetative Growth And The P And K Uptake Of Okra Abelmoschus Esculentus Seedling
Use mouse wheel to zoom in and out Drag zoomed map to pan it Double‑click a ticker to display detailed information in a new window Hover mouse cursor over a ticker to see its main competitors in a stacked view with a 3month history graphThe explanation is relatively straightforward An sp orbital is composed of one s orbital and one p orbital, and thus it has 50% s character and 50% p character sp 2 orbitals, by comparison, have 33% s character and 67% p character, while sp 3 orbitals have 25% s character and 75% p The S&P 500 Dividend Yield, as calculated by the S&P 500 Dividends Per share TTM divided by the S&P 500 close price for the month, reflects the dividendonly return on the S&P 500 index The S&P 500 index is a basket of 500 large US stocks, weighted by market cap, and is the most widely followed index representing the US stock market



Plos One P Coumaric Acid Influenced Cucumber Rhizosphere Soil Microbial Communities And The Growth Of Fusarium Oxysporum F Sp Cucumerinum Owen




Expression Of Substance P Sp In The Human Adrenal Gland A Download Scientific Diagram
P orbital is an atomic orbital having a dumbbell shape P orbitals have a higher energy than that of s orbitals The letter "p" stands for "principal" It describes the angular momentum of electrons in the p orbital One p orbital can hold a maximum of 6 electrons These electrons occupy subatomic orbitalsMy Account TSP Account Number User ID Forgot your account number or user ID? In an #sp^3# hybridization, #color(red)"one"# #s# orbital is mixed with #color(red)"three"# #p# orbitals to form #color(red)"four"# #sp^3# hybridized orbitals Each of these hybridized orbitals have 25% s character and 75% p character (calculated according to the proportion of sp mixing) These #sp^3# hybridized orbitals are oriented with bond angle of




Alterations Of Substance P Sp Levels At Superficial Laminae Of L5 S2 Download Scientific Diagram




Audiosquare Fujisawa 話題の小型スピーカー Celsus Sound セルサス サウンド Sp One P を期間限定でお聴き頂けます
Sony Original Playstation One Console (Renewed) Amazon Renewed 43 out of 5 stars 273 PlayStation 5 offers from $ Playstation Classic Console with Classic Playstation Games PreInstalled Holiday Bundle, Includes Final Fantasy VII, Grand Theft Auto, Resident Evil Director's Cut and More SonyAn sp^3 orbital is unsymmetrical in shape, having one small and one large lobeThe four sp^3 hybrid orbitals of a group are equivalent in shape and energy Select all the statements that correctly describe hybridization involving d orbitalsthe hybridization of one s, three p and one d orbital gives five sp3d orbitalsESE Policies & Procedures (SP&P) Section (1) (b)1, Florida Statutes (FS), requires that district school boards submit to the Florida Department of Education (FDOE) proposed procedures for the provision of special instruction and services for exceptional students once every three years Approval of this document by FDOE is required by



Boston 2crsi Octopus 1 2sp One Node With Two Gpus



Plos One The Novel Reversible Lsd1 Inhibitor Sp 2577 Promotes Anti Tumor Immunity In Switch Sucrose Nonfermentable Swi Snf Complex Mutated Ovarian Cancer
Latest News See all of our latest news Laundry Done Right Malcolm Gladwell Explains Cold Water Wash Read more P&G Ventures Among Top Best Workplaces for Innovators Read more Engage, Act, and make an Impact Read more Delivering on Diversity Read more Our brandsRecent awards include Best Credit Risk Management Product; S&P 500 P/E Ratio Forward Estimate is at a current level of 2194, down from 2267 last quarter and down from 2372 one year ago This is a change of 3% from last quarter and 750% from one year ago



Plos One Seed Biopriming With P And K Solubilizing Enterobacter Hormaechei Sp Improves The Early Vegetative Growth And The P And K Uptake Of Okra Abelmoschus Esculentus Seedling



One Hop Screen Room One Hop Throughput Comparison Sp Vs Ss Download Scientific Diagram




Spone



Gray Anchor Roma 10a S P One Way Switch Module Size 1 Module 230 V Rs 30 Piece Id




How Many Pi Bonds Can An Sp Atom Form Homeworklib




Sp3 Sp2 And Sp Hybridization In Organic Chemistry With Practice Problems Chemistry Steps




Bel Artsp Scienceware Single Piece Buchner Funnels 400ml Capacity 104mm Fisher Scientific



More Marineford Characters Join One Piece Unlimited Cruise Sp Siliconera




One Hit Destruction Vegeta Sp Set Promo P 001 Pr Dragon Ball Super Singles Dragon Ball Super Promotion Cards Coretcg



キャンペーン 新品未使用 Celsus Sound Sp One P パッシブタイプ 2本1組 スピーカー セルサスサウンド Spone P 通販 Lineポイント最大0 5 Get Lineショッピング




Two Oocysts Of Eimeria Sp One Viable And Sporulating On The Left Download Scientific Diagram




Solved Which One Of The Following Statements Is False A An Chegg Com




Skirted One Piece Dual Flush 3 4 5l Washdown Toilet With P Trap 453x Sp Kohler
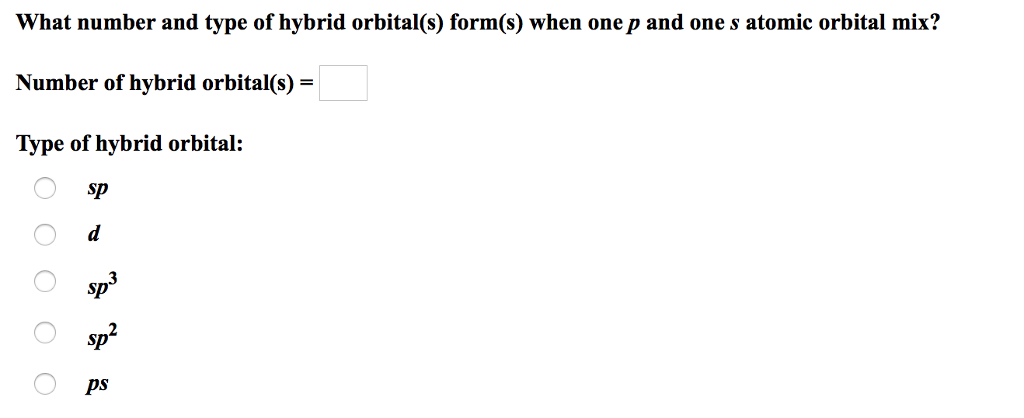



Solved What Number And Type Of Hybrid Orbital S Form S Chegg Com




Hybridorbitals 01 9701 A Level Chemistry Studocu




Delaware S P Ebt Program Will Provide One Time Benefits To Feed Children During Summer State Of Delaware News




Astell Kern Acro L1000 Celsus Sound Sp One P Kupit V Kieve I Ukraine Po Cene Grn Na Portativ Ua
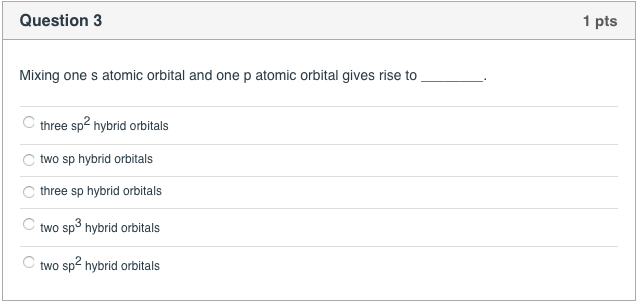



Solved Question 3 1 Pts Mixing One S Atomic Orbital And One Chegg Com



Whats Coming Up Oct 25 Oct 27 Oct




Hybrid Orbitals Youtube




The Blending Of One S Atomic Orbital Three P Atomic Chegg Com




Amazon Com Salt And Pepper Shaker Set Of 2 With Stainless Steel Lids Hand Painted Distressed Kitchen Table Decor White Gray Seafoam Teal Blue Green Cream Brown Tan



Chapter 7 Chemical Bonding Ppt Download




Organic Chemistry Organic Chemistry The Study Of The Compounds Of Carbon Over 10 Million Organic Compounds Have Been Identified About 1000 New Ones Ppt Download




Alterations Of Substance P Sp Levels At Superficial Laminae Of L5 S2 Download Scientific Diagram



One Piece P O P Limited Edition Nami Ver Sp Figure Megahouse 100 Authentic




Sp Monogram Logo Monogram Logo Letters Monogram Logo S Letter Images




Sch4u1 September Mr Dvorsky Ppt Download



Chapter 2



Answer With Counter Claim S Jam Pharmacy Corp Trellis



Lecture 16 C 1403 October 31 05 18




Hybridization Mixing Of Two Or More Atomic Orbitals



Sigma Bonds From Sp And Sp2 Hybrid Orbitals




The Echo Opera In One Act Lt Sp 25 3 39i O S Si Prprpr R Rp Rpr Prp Rpr Prp Rfr 4 S I I4i J St 1 2



Plos One The Novel Reversible Lsd1 Inhibitor Sp 2577 Promotes Anti Tumor Immunity In Switch Sucrose Nonfermentable Swi Snf Complex Mutated Ovarian Cancer



One Piece P O P Limited Edition Nami Ver Sp Figure Megahouse 100 Authentic
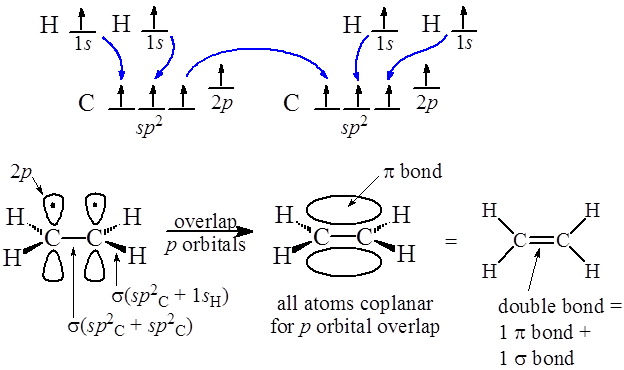



Molecular Formulas And Nomenclature
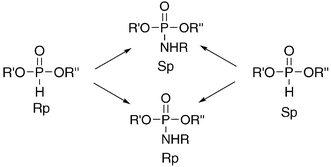



Controlling Stereochemistry During Oxidative Coupling Preparation Of Rp Or Sp Phosphoramidates From One P Chiral Precursor Chemical Communications Rsc Publishing




Monkey Salt And Pepper Balancing Act One Pc Set Monkey S P
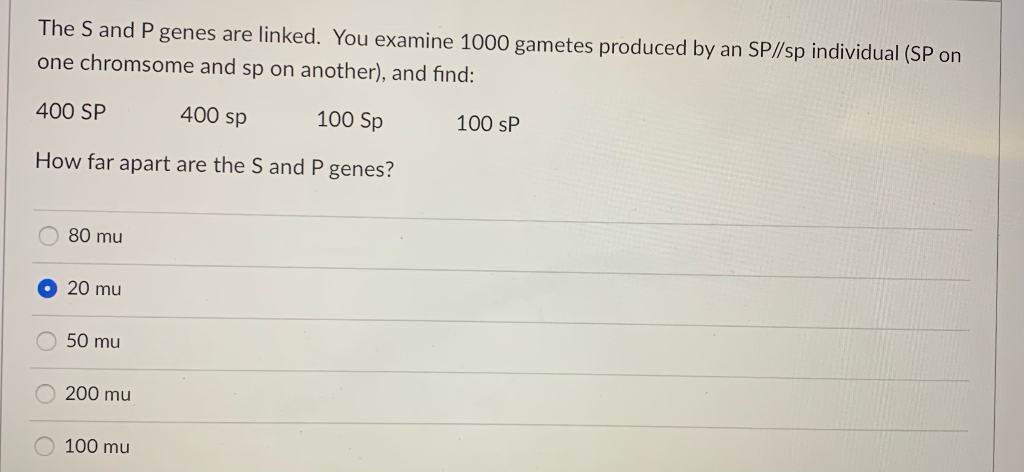



The S And P Genes Are Linked You Examine 1000 Chegg Com




One Hybridization Of One S And One P Orbital We Get




Arena Sp Der One P Ece Girls Swimwear Superdokan
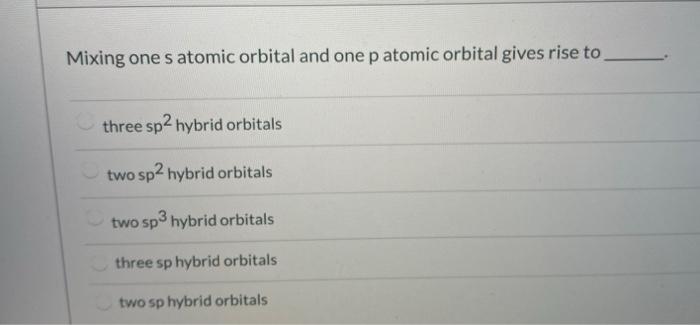



Mixing One S Atomic Orbital And One P Atomic Orbital Chegg Com




Complete Library Of Cookery Three Volumes In One Lee Mrs N K M Mis Eliza Leslie S P Hedges Md Mrs Emma P Ewing Amazon Com Books




Celsus Sp One Active Speakers Review Headfonics Com




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase




One Piece 937 Manga One Piece 937 English One Piece 937 Read Read One Piece 937 Manga One Piece 937 Sub English One P One Piece Chapter Manga English One Piece




P S Rcd113 3 Switch Single Pole 3 Way Sp Sp Gordon Electric Supply Inc



Hybridization Department Of Chemistry



Apple Gate Project Bushwick Gorey Sp One Whisper Quik Binho
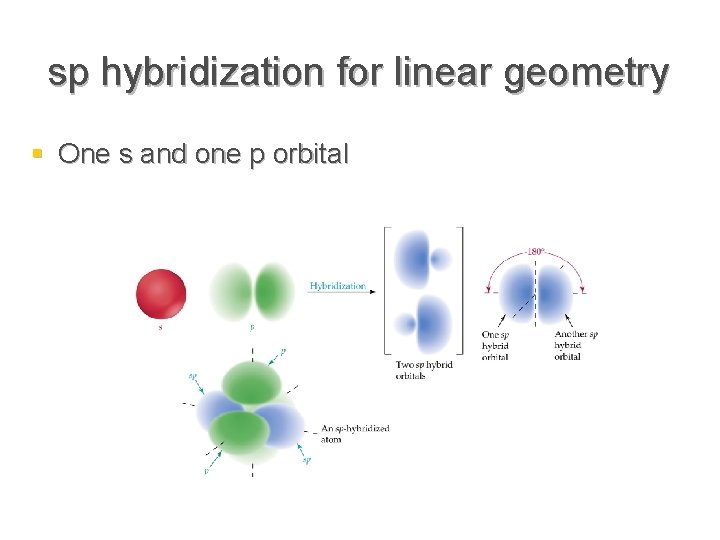



Valence Bond Theory Electrons Are Not Simply Dots




Introduction To Bonding Ppt Download



Plos One Seed Biopriming With P And K Solubilizing Enterobacter Hormaechei Sp Improves The Early Vegetative Growth And The P And K Uptake Of Okra Abelmoschus Esculentus Seedling



Mixed Strategy Nash Equilibrium For The One Shot S P Game Download Scientific Diagram




Megahouse P O P X Pinkyst Street Nami One Piece Figure




Simple Plan Su Twitter Vip Pizza Party Upgrades For The Pop Punk S Still Not Dead Tour Are On Sale Now At T Co Nqczbtj19z We Re So Excited To Finally Be Back On Stage Playing




Liz Lange Swim New Liz Lange Maternity Sz Sp One Piece Swim Suit Poshmark
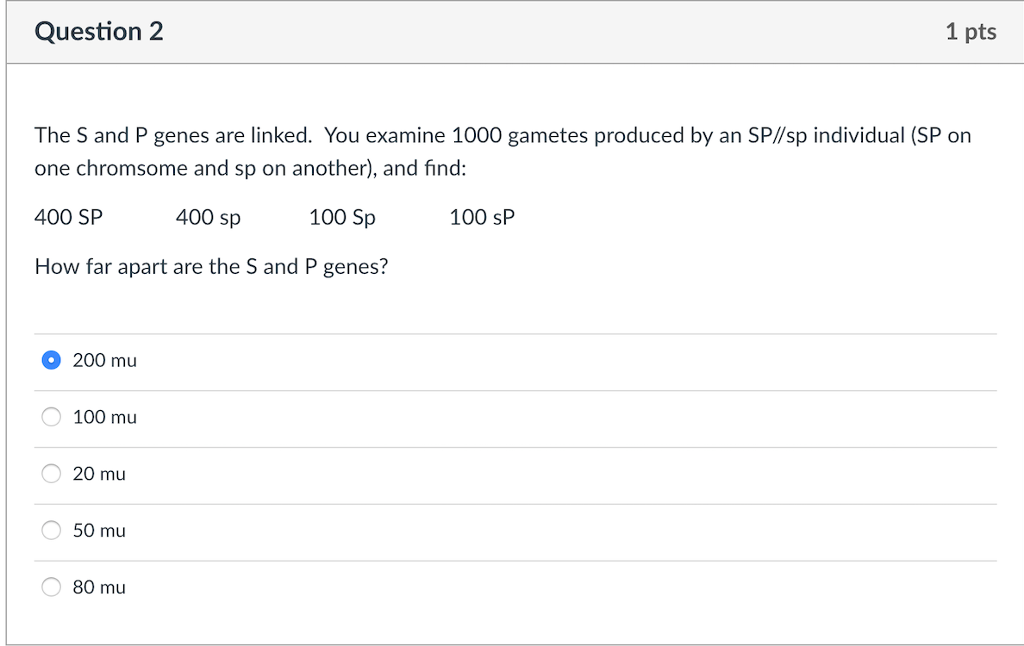



Solved Question 2 1 Pts The S And P Genes Are Linked You Chegg Com




Amazon Com Roland Xc 540 Sc 540 Sp 300 P Roller Td16s4 Type2 One Couple Us Stock Office Products




A Sp Hybridisation One S Orbital Mixes With One P Orbital Forming Two Equivalent Sp Hybrid Orbitals Types Of Hybridisation Linear 180 Two Sp Hybrid




Snowdrop Galanthus Sp Single Flower In Snow Print




Annotationes Zoologicae Japonenses Nihon Do Butsugaku Iho New Or Imperfectly Known Species Of Earthworms 15 P Shimaensis N Sp One Specimen Preserved In Formol Length 5 Mm Breadth 7 Mm




Spone



Plos One P Coumaric Acid Influenced Cucumber Rhizosphere Soil Microbial Communities And The Growth Of Fusarium Oxysporum F Sp Cucumerinum Owen




One Sp Controls 501 001spc Sp2 Chassis Pcb Working For Sale Online Ebay
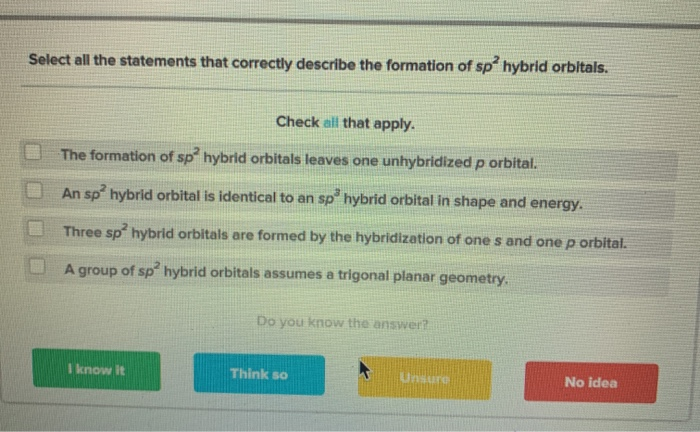



Solved Select All The Statements That Correctly Describe The Chegg Com




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase




Sp One P Celsus Sound Hifi Do Mcintosh Jbl Audio Technica Jeff Rowland Accuphase



Baby Silicone Plate Bibs Bowl Spone Feeding Tableware Waterproof Non Slip Crockery Bpa Free Silicone Dishes For Baby Bowl Baby P Dishes Aliexpress




Porcelanosa Sp One Xl Bathtub Tiles And Bathrooms Online
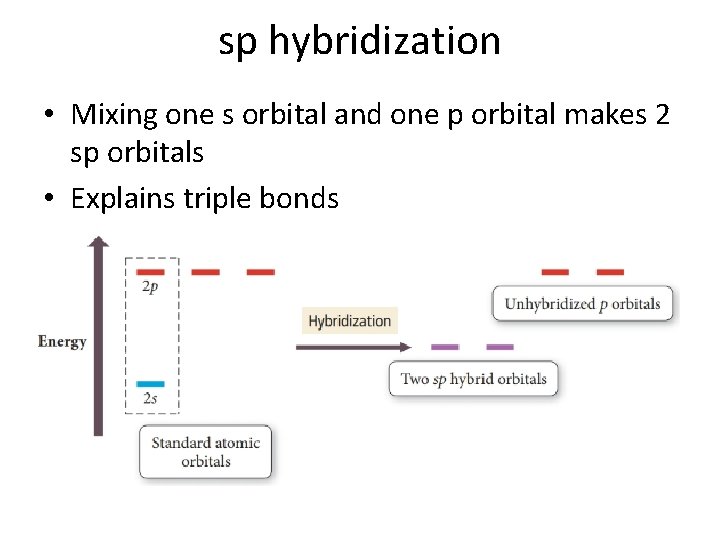



Chapter 10 Chemical Bonding Molecular Shapes Valence Bond




One Piece T Shirt Men Unlimited Cruise Sp Japan Anime The Straw Hat Pirates Luffy Tshirt Summer Cotton Shirts Black Short Sleeve T Shirts Aliexpress



コメント
コメントを投稿